Electrochemical Techniques
We can place the most popular voltammetric techniques into three general categories:
- Pulse (or step) techniques,
- Sweep techniques, and
- Hydrodynamic techniques.
Most of the electrochemical techniques within a given category are very similar. For all of these,
the potential of the electrode is varied reproducibly, so that the analyte is either
oxidized or reduced at the surface of the electrode, creating a concentration gradient, and hence a current response. Sweep techniques are preferred for studying the overall electrochemical behavior of analytes in a solution, while step techniques generally give better quantitative information. Hydrodynamic techniques involve stirring the electrochemical solution in order to control the concentration gradient.
Techniques:
- Cyclic Voltammetry
- Chronoamperometry/Chronocoulometry
- Pulse Voltammetry
- Stripping Voltammetry
- Digital Simulation
- Microelectrodes
- Glucose Detection
- Electrochemical Polymerization
- Electrochemical Impedance Spectroscopy
- Corrosion
Below is a chart depicting the relation between many various types of electroanalytical techniques.
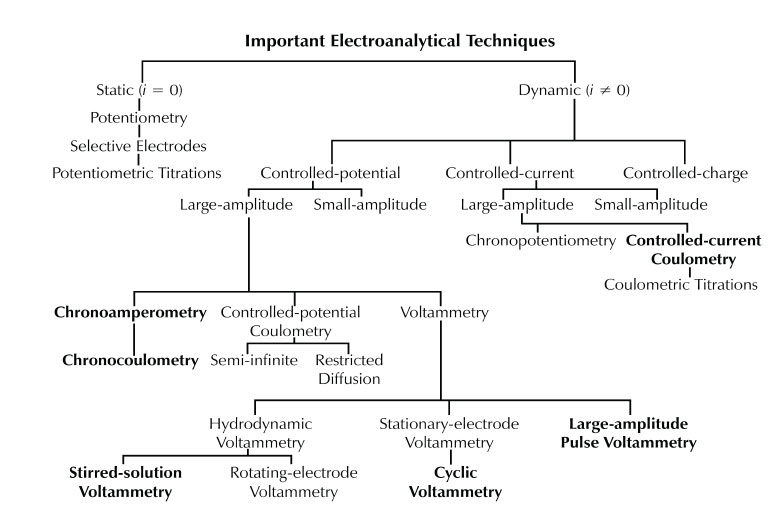
Adapted from P.T. Kissinger and W.R. Heineman, Laboratory Techniques in Electroanalytical Chemistry, New York: Marcel Dekker, 1984, p. 6.)
Techniques used in this course are shown in boldface type in this chart.
Controlled-potential measurements are called potentiometric; controlled-current measurements are called galvanometric. Potentiostatic measurements hold the voltage in a system constant; galvanostatic measurements hold the current in a system constant.
