Electrochemical Lab
Manual Equations
- Anson Equation
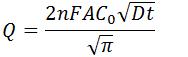
- Q is charge (C)
- n is number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- A is surface area of the working electrode (cm2)
- C0 is initial concentration of the analyte (mol/cm3)
- D is diffusion coefficient of the analyte (cm2/s)
- t is time (s)
- Corrosion Rate
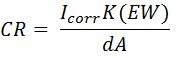
- CR is corrosion rate in milli-inches per year (mpy)
- Icorr is corrosion current (A)
- K is constant that defines the units for corrosion (1.288 x 105 milli-inches/A·cm·year)
- EW is equivalent weight (g/equivalent)
- d is density (g/cm3)
- A is area of the electrode (cm2)
- Cottrell Equation
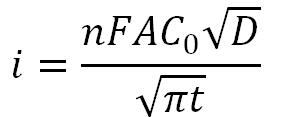
- i is current (A)
- n is number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- A is surface area of the working electrode (cm2)
- C0 is initial concentration of the analyte (mol/cm3)
- D is diffusion coefficient of the analyte (cm2/s)
- t is time (s)
- Diffusion Layer Thickness
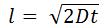
- l is distance (cm)
- D is diffusion coefficient of the analyte (cm2/s)
- t is time (s)
- Faraday’s Law of Electrolysis
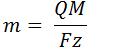
- m is mass (g)
- Q is charge (C)
- M is molar mass (g/mol)
- F is Faraday’s Constant (96,485.3 C/mol)
- z is valance number
- Fick’s First Law of Diffusion
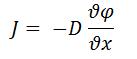
- J is diffusional flux (mol/m2s)
- D is diffusion coefficient of the analyte (m2/s)
is concentration (mol/m3)
- x is position (m)
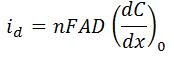
- id is diffusion limited current (A)
- n is the number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- A is surface area of the working electrode (cm2)
- D is diffusion coefficient of the analyte (cm2/s)
- (dC/dx)0 is concentration gradient at the surface of the electrode
- Formal Peak Potential
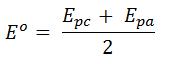
- Eo is the formal potential of the analyte
- Epc is the cathodic peak potential
- Epa is the anodic peak potential
- Levich Equation
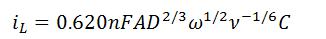
- iL is Levich current (A)
- n is the number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- A is surface area of the working electrode (cm2)
- D is diffusion coefficient of the analyte (cm2/s)
is the angular rotation rate of the electrode (rad/s)
- ν is the kinematic viscosity (cm2/s)
- C is concentration of the analyte (mol/cm3)
- Nernst Equation
-
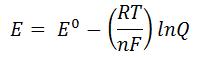
-
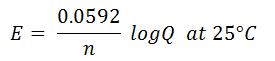
- E is cell potential at the temperature of interest
- Eo is the standard cell potential
- R is the Molar Gas Constant (8.31446 J/mol•K)
- T is temperature (K)
- n is number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- Q is the reaction quotient
-
- Ohm’s Law
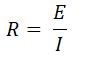
- R is resistance
- E is applied potential
- I is applied current
- Peak-to-peak separation for reversible reaction
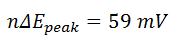
- n is the number of electrons transferred
Epeak is the different between the cathodic and anodic peak potentials
- Randles – Sevcik equation
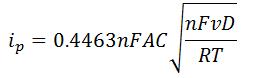
-
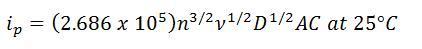
- n is the number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- A is surface area of the working electrode (cm2)
- D is diffusion coefficient of the analyte (cm2/s)
- R is the Molar Gas Constant (8.31446 J/mol•K)
- T is temperature (K)
- Ratio of peak heights for a reversible reaction
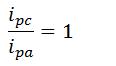
- ipc is cathodic peak current
- ipa is anodic peak current
- Steady-State Current at a Microelectrode
-
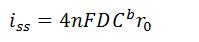
- I is steady-state current (A)
- n is the number of electrons transferred
- F is Faraday’s Constant (96,485.3 C/mol)
- D is diffusion coefficient of the analyte (cm2/s)
- Cb is analyte concentration (mol/cm3)
- r0 is radius of the electrode (cm)
-
- Stern Geary Equation
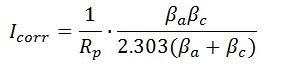
- Icorr is corrosion current (A),
- Rp is polarization resistance
- βa is anodic Tafel constant
- βc is cathodic Tafel constant
