Determination of Acetaminophen
Goal
- To determine the concentration of acetaminophen in a commercial pain-reliever of two forms, tablet and elixir.
Experimental apparatus
- Gamry Instruments Interface 1000T potentiostat
- Gamry Instruments Framework™ software package installed on a host computer
- Screen-printed electrode cell stand (990-00420)
- Platinum working screen-printed electrode (935-00122)
- Volumetric flasks (one 1 L, two 100 mL, six 25 mL)
- Volumetric pipettes (0.5, 1.0, and 2.0 mL)
Reagents and chemicals
- Acetaminophen stock solution
- Potassium chloride
- Sodium hydrogen phosphate heptahydrate
- Citric acid monohydrate
- Commercial acetaminophen elixir
- Commercial acetaminophen tablet
Background
The major active ingredient in several pain relief medicines is 4-acetamindophenol,
or commonly called acetaminophen or paracetamol. It comes in a few different formats for consumers such as tablet, capsule, and liquid. This experiment is designed to determine the amount of acetominophen in one dose of medicine using cyclic voltammetry. The result is compared to the stated concentration as per the manufacturer.
Acetaminophen is one of the most popular pain relievers for general ailments. Depending upon the tablet or capsule, a typical dose can range from 80 mg (children’s dose), to 650 mg (adults’ extended-relief capsules). In the liquid form, doses can vary from 16 mg/mL to 22 mg/mL.
Acetaminophen undergoes an irreversible electrochemical reaction at the face of an
electrode. (An example of a reversible reaction would be the first experiment in this
book, in which cyclic voltammograms were performed on potassium ferricyanide.)
Unlike ferricyanide, acetaminophen is oxidized via a two-electron process. This oxidized acetaminophen is then chemically converted in aqueous acid to the hydrated form of N-acetyl-4-quinoneimine, as shown in the reaction below.
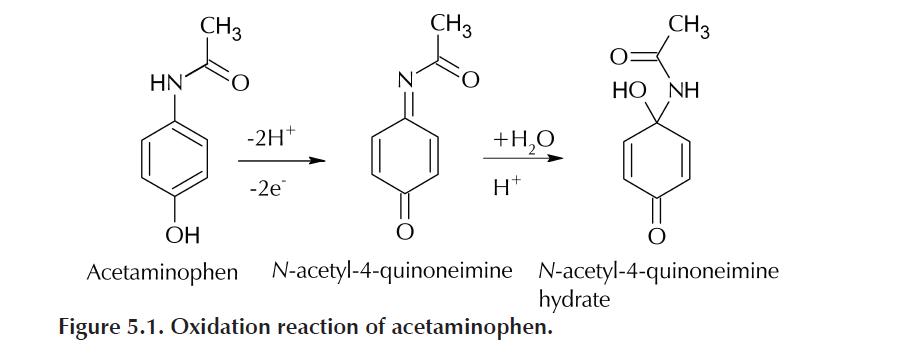
The reverse of this chemical reaction is not kinetically favored, and therefore the
electrochemical reaction of acetaminophen is essentially irreversible. This type of
reaction, in which an electrochemical reaction is followed by a chemical reaction, is
commonly known as an EC reaction or EC mechanism. (We discuss mechanisms more in the next laboratory experiment with simulations.)
In the cyclic voltammogram for this process, there is a peak in the forward scan, but there is no cathodic peak when the potential scan is reversed. This is because the oxidized form of acetaminophen has undergone a chemical reaction, and it is not present to be reduced. This leads to the observation of an unusual voltammogram. The anodic peak however can still be used in the determination of acetaminophen’s concentration because the forward scan is unaffected by this chemical reaction.
